Products for Development for Additive Manufacturing
Projet 7000 Hd
SLA の品質と精度を実現するミドルクラスの統合ソリューション
Figure 4 Standalone
即日プロトタイピングや少量生産を高速に低コストで実現
SLS 380
選択的レーザー焼結向けの工業スケールのプロセス制御された積層造形ソリューション
ProJet MJP 2500/2500 Plus
マルチジェットプリントで品質、スピード、使いやすさを実現


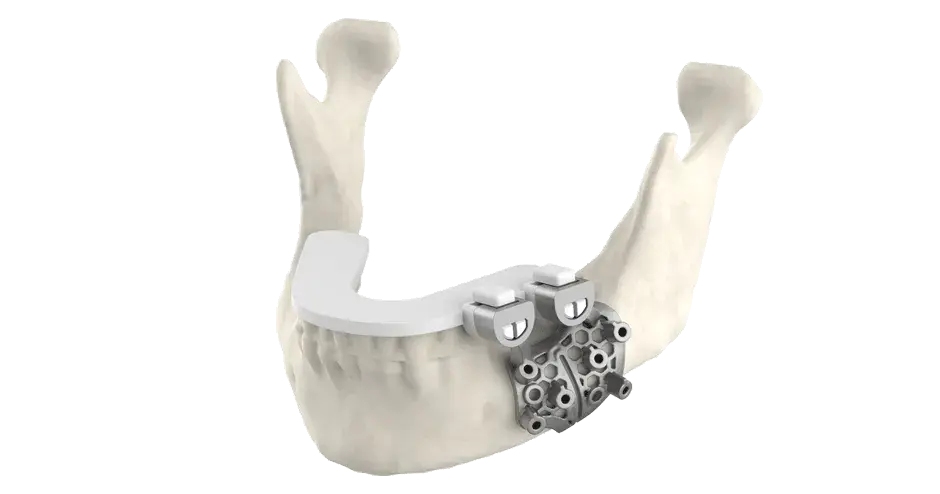
NuVasive Taps AM Ecosystem to Optimize Spine Implant Technology
Medical device manufacturers need innovative ways to bring high-quality, reliable products to market faster. Our customers trust our unparalleled capabilities and experience in additive manufacturing (AM) to help them develop innovative medical devices and surgical instruments. We also collaborate with them to determine the best approach to commercialization, either through contract manufacturing support from our two device manufacturing facilities or via technology transfer to our customer.
Our experienced team can help speed time to market and remove risk from the device development process by combining our validation strategy for Direct Metal Printing (DMP) and leveraging process capability with titanium alloy Ti6Al4V. Discover how.

Whether you are representing an industry leader or an innovative startup, 3D Systems can help you develop groundbreaking precision healthcare applications and medical technologies. Our experience with regulatory and compliance processes enables us to help our customers bring revolutionary 3D printed medical devices to market faster than ever before.

In fact, our development team is continuously innovating our printers, software, and processes to make the manufacture and commercialization of surgical implants and instruments, quicker and more cost effective. The ProX® DMP 350 and 500 platforms are setting the benchmark in material properties and surface resolution for metal parts. Our 3DXpert™ software helps you design devices specifically for AM. It even provides the ability to create complex porous structures, which typically could not be created through traditional subtractive manufacturing.

Our Application Innovation Group (AIG) is a team of engineers, designers, and technicians who live and breathe AM. We work closely with medical device manufacturers, collaborating with them from concept to commercialization of 3D printed implants and instruments within a variety of surgical specialties.
Together we’ll identify your needs, work with you to optimize your designs, and define manufacturing flow to achieve your application's requirements. Then, we’ll move you toward certification and validation while assisting with obtaining regulatory clearances, completing technology transfer, and implementing AM production on-site or within one of our facilities.

With experience manufacturing over two million medical devices, beginning with the very first FDA-cleared 3D printed titanium implant, 3D Systems has world-class, FDA-registered, ISO 13485-certified facilities. These advanced, highly-productive facilities—located in both North America and Europe—have become a preferred choice among medical device manufacturers.
Additive manufactured implants and surgical instruments exhibit excellent mechanical properties enabling accelerated product development and lower inventory costs compared to traditional subtractive manufacturing and its long lead times. Moreover, validated parameter sets enable printing of metallic parts that meet ASTM standards for mechanical properties and chemical composition. The 3D printing process also yields many cost-effective implant types and is fully compatible with traditional (CNC) manufacturing.
3D Systems is the trusted partner of top medical device OEM’s to manufacture their implants and instruments for spine, orthopaedic, extremity, craniomaxiofacial applications and more. Contact us to start your next project.






Discover how our healthcare materials respond to autoclave, EtO, novel gas, gamma, and e-beam methods of sterilization, enabling you to classify materials as production-capable, clinical trial-capable, or appropriate for functional prototyping. Find out more.
The materials used for personalized healthcare solutions have gone through extensive quality and regulatory testing in order to ensure they meet the needs of 3D Systems healthcare solutions team and our customers. Learn more about biocompatible materials that can be sterilized and used to guide osteotomies as well as non-sterilizable materials for pre-surgical planning and education.
SLA の品質と精度を実現するミドルクラスの統合ソリューション
即日プロトタイピングや少量生産を高速に低コストで実現
選択的レーザー焼結向けの工業スケールのプロセス制御された積層造形ソリューション
マルチジェットプリントで品質、スピード、使いやすさを実現